Why Is Biopharmaceutical Innovation Important?
New and innovative medicines bring demonstrable value to patients, our health care system, and society by:
- Better targeting diseases, developing personalized therapies, and improving treatment protocols that help to reduce patients’ side-effects and adverse events; and
- Keeping patients out of the hospital and avoiding other costly inpatient expenditures, which helps to reduce downstream costs to our entire health care system.
The increasing prevalence of chronic illnesses and conditions, particularly among older Americans, underscores the importance of continued medical innovation aimed at preventing, treating, and curing these costly, life-threatening diseases. Over the past decade, growing numbers of patients and families have benefited enormously from innovative new medicines that address an ever wider range of acute and chronic diseases. With patients in mind, support for innovation and discovery is critical to confronting the projected escalation of complex conditions like Alzheimer’s Disease, cancer, and diabetes.
Understanding the Current Policy Climate
To encourage these discoveries, it is important to understand the kind of climate and incentives for innovation and how these incentives may be affected by a variety of public policies. There are many concerns about patient and health system costs and accessibility to treatments, as well as how certain decisions – or trade-offs – may impact overall spending and patient care.
It’s also important not to view the cost of drug development in a vacuum, but rather the totality of these costs within the context of both time (the extent to which drug development can slow the escalation of, and the future costs associated with, chronic disease, as well as the inevitable decreasing of drug costs when competitors enter a therapeutic category’s marketplace) and space (the role of pharmaceutical innovation within a health care system in the process of transformation).
As part of the conversation about the cost of new cures and treatments, it’s essential to include a discussion about how the U.S. health system could be made much more cost-efficient by eliminating hundreds of billions of dollars in waste. Studies have shown that, at a minimum, one of every five health care dollars is lost to overtreatment, breakdowns in care coordination and other system failures that stand to be repaired as the nation transitions to a pay-for-value approach to care delivery.
Among the questions that need to be considered by stakeholders:
- How do we put all health care costs on the table to determine where we get the best long-term value in health care for patients, the economy and society?
- Given the unique nature of the life cycle of pharmaceutical innovations, are there methods or tools that should be utilized, or developed, to weigh the potential long-term health savings generated by a new therapy versus its immediate costs?
- Within health care payment systems, should policy-makers encourage innovative risk mechanisms that would incentivize and optimize impactful drug development?
This is a discussion that will profoundly affect our nation’s health and future, and one that the National Pharmaceutical Council is working to continue through our research and collaboration with other health care stakeholders.
Impact on Outcomes and Spending
For decades, biopharmaceutical products have been an important tool in achieving positive health outcomes for patients with a variety of conditions.
NPC research found investments made 20 years ago to combat the worst diseases of that period are continuing to pay off. Six of the top seven causes of death and disability in 1995 have seen significant improvements in patient outcomes associated with these investments. Findings from a physician survey indicated that pharmaceutical and biopharmaceutical products were viewed as accounting for the greatest impact on mortality and morbidity across these top conditions. And biopharmaceuticals developed in that period and beyond represent significant advances in treatment for diseases and conditions such as cancer, cerebrovascular diseases, HIV and others.
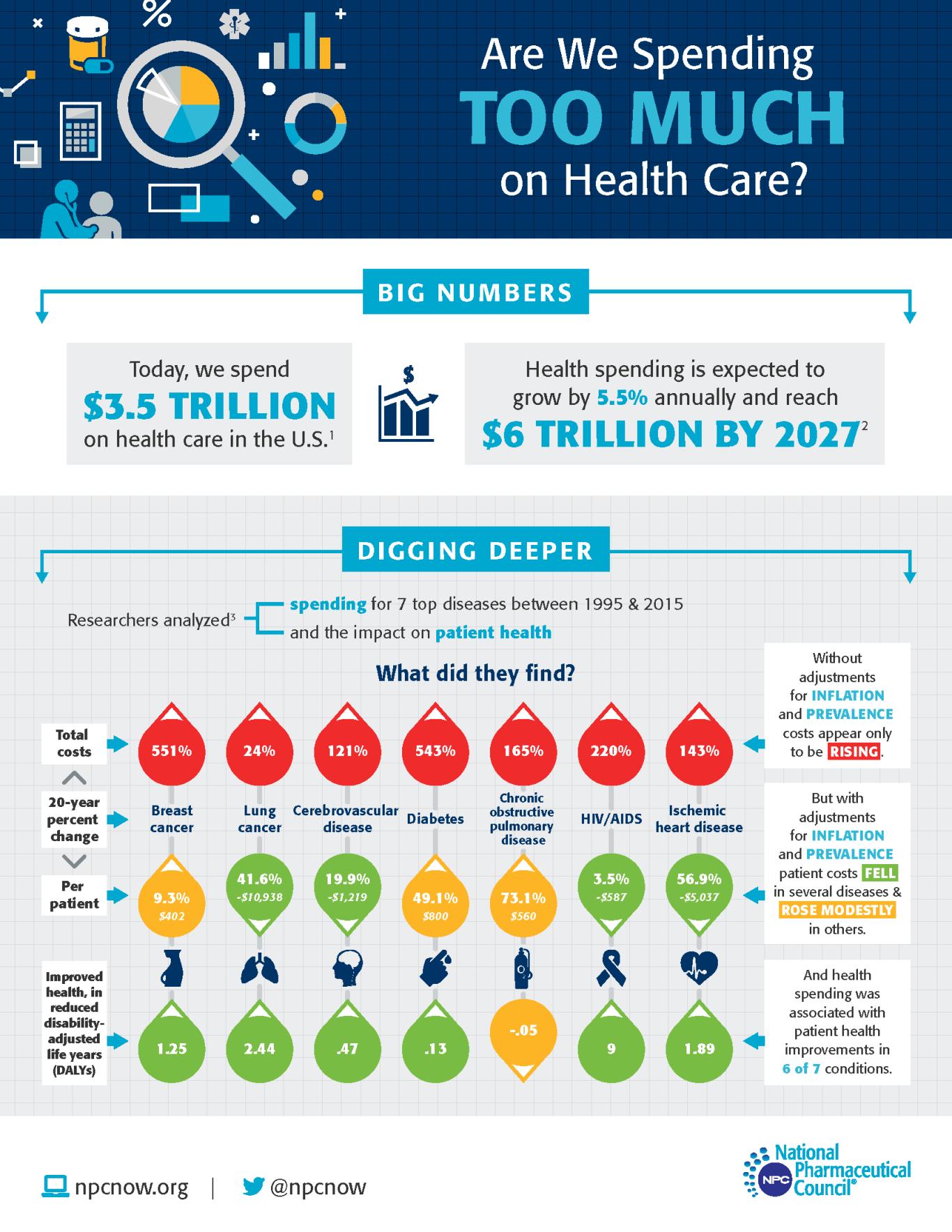
These findings were reinforced by other researchers who quantified how public health, pharmaceutical products and other medical care contributed to improved life expectancy between 1990 and 2015. The research found that 35% of the increase in life expectancy was attributable to biopharmaceuticals. In comparison, other medical care accounted for only 13%.
Beyond life expectancy, other NPC research found that novel specialty therapies slow disease progression and improve patient quality of life for conditions with high specialty pharmaceutical spending and use of Medicare Part B products for rheumatoid arthritis, multiple sclerosis and breast cancer.
The ongoing COVID-19 pandemic underscores the need to consider policies that promote innovation as researchers across the globe work to develop therapeutics and vaccines to combat this virus. In totality, these research findings demonstrate how vital pharmaceutical products are and the need for policies that recognize their benefits and encourage, not dampen, future investment in innovations that will lead to improved patient outcomes in the decades to come.
What's been the bang for the buck in health care spending?
This study assessed whether increased medical intervention spending on prevalent chronic conditions has been a good investment over time.
